Northern Lights visible in UK skies

The week which marked the end of February and the start of March brought along not one but two fun astronomical spectacles: the planet conjunction and the northern lights. In this article, Almudena Visser Velez explains the phenomenon of the northern lights.
The northern lights, or aurora borealis, are perhaps one of the most beautiful spectacles astrophysics has to offer, and some parts of the UK were unusually lucky to witness illuminated skies and dancing colours at the beginning of March. Typically a phenomenon associated with the north and south poles, space weather can result in the auroras extending away from the poles, often reaching parts of Scandinavia in the northern hemisphere but rarely coming as far as the UK. While they are extraordinary to look at, the study of auroras is important scientifically too as it helps us understand complex space weather and its impact on human technology.
Solar wind, a constant stream of charged particles coming from the Sun, flows outwards in all directions, with particles travelling millions of kilometres per hour. A tiny fraction of these high speed particles reach Earth and when they do so they react with our magnetic field. This spherical field, also called the magnetosphere, extends several tens of thousands of kilometres into space and helps protect Earth from high energy particles such as these which constantly bombard us and other planets. Once captured by the magnetic field, the particles are accelerated towards the poles where the field is strongest, which explains why these areas are most likely to witness aurora spectacles.
Typically a phenomenon associated with the north and south poles, space weather can result in the auroras extending away from the poles, often reaching parts of Scandinavia in the northern hemisphere but rarely coming as far as the UK.
The flashing lights are caused by the charged particles slamming into molecules and atoms in our atmosphere, kicking atomic shell electrons into higher energy levels in a process called ‘excitation’. Sufficiently energetic incoming particles may give the electron so much energy that the result is ‘ionisation’, where electrons are booted out of atoms completely and the atom becomes more positively charged, which readers may remember from school chemistry lessons. The extra energy provided to the atmospheric molecules is released as photons (particles of light) to allow the electrons to return to their favoured low energy ‘ground state’. Auroras usually come in green or pink hues and the various colours are caused by the different chemicals in our atmosphere, in this case oxygen and nitrogen respectively. A sketch of the process can be found below.
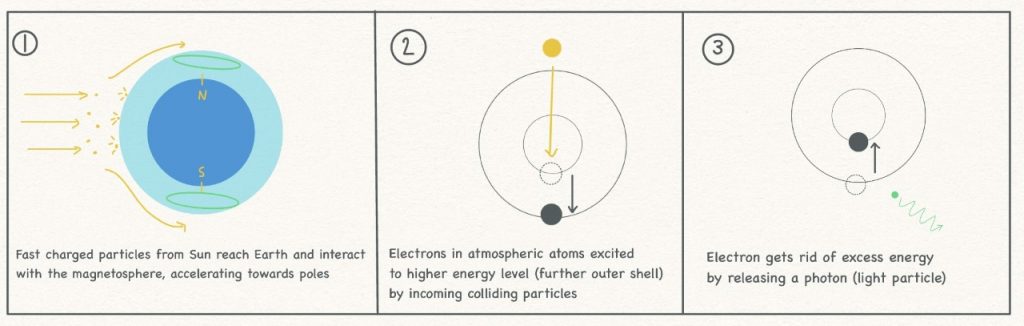
The lights have different colours as the wavelengths of the photons released to return the energy of the excited electron to the ground state varies in different chemicals owing to different electronic structures. What we see in the sky is the result of billions of individual particle interactions on a large scale.


